![]()
![]()
1/2 /3-4/ 5-9/Aa-Am/An-Az/B/Ca-Ce/Cf-Cz/Da-Dh/Di-Dz/E/F/G/H/I/J/K/L/M/N/O/Pa-Pf/ /Pj-Po/Pp-Pz/Q/R/Sa-Ss/St-y/Ta-Th/Ti-Tz/U/V/W/X/Y/Z / Chinese spelling
|
|
|
offer you better
services
|
|||||||||||||||||||||||||||||||||||||||||
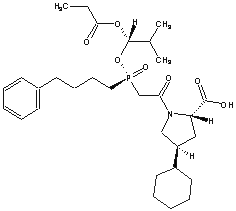
| FOSINOPRIL SODIUM | |||||||||||||||||||||||||||||||||||||||||||
|
PRODUCT:Fosinopril
Sodium
|
|||||||||||||||||||||||||||||||||||||||||||
| KEY INTERMEDIATES | |||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||
| Applications | |||||||||||||||||||||||||||||||||||||||||||
| Fosinopril
is in a class of drugs called ace inhibitors which inhibit ace and
block the formation of angiotensin ii. It is used in the treatment
of high blood pressure and congestive heart failure. more.... |
|||||||||||||||||||||||||||||||||||||||||||
| Packing &loading | |||||||||||||||||||||||||||||||||||||||||||
| This data is only for your information purposes and does not imply guarantee for a certain application. | |||||||||||||||||||||||||||||||||||||||||||
| If you need an indicative price to your country, please kindly advise the annual quantity needed, destination port and we will send you our soonest CNF(Cost + Freight) price. Your indication for a workable price level will sure speed up our fastest service for you | |||||||||||||||||||||||||||||||||||||||||||
|
We really need to get
feed back from our customers in order to improve our performances. Best regards from SHENZHEN , China Mr. JEFFERY TSUI |
|||||||||||||||||||||||||||||||||||||||||||
| Material Safety Data Sheet | |||||||||||||||||||||||||||||||||||||||||||
| Related Information and materials | |||||||||||||||||||||||||||||||||||||||||||
fosinopril sodium Main Index |
|||||||||||||||||||||||||||||||||||||||||||
| If the above information doesn't cover |
contact us
|
||||||||||||||||||||||||||||||||||||||||||
![]()
![]()